|
All 275 seropositive sera detected by the Focus, Kalon, and Biokit assays were confirmed using the University of Washington gold standard Western Blot Assay 2 University of Washington, Western Blot Assay. The pre-market Biokit assay showed 96% sensitivities and 98% specificities. The Biokit assay is a point-of-care test and was developed as a rapid membrane assay for detecting HSV-2 antibodies. Kalon had a sensitivity/specificity of 100% each. It is used mainly in the United Kingdom and Europe. The Kalon kit is not FDA approved (as of September of 2015) and is used primarily in secondary research settings in the United States. The kit has been modified to eliminate any reactivity from HSV-1 infections while being able to detect antibodies against HSV-2. Any anti-HSV-2 IgG in the sample is detected in a photometer. Kalon is an ELISA assay, which uses 96 well microtiter plates coated with a recombinant gG2 antigen for HSV-2. 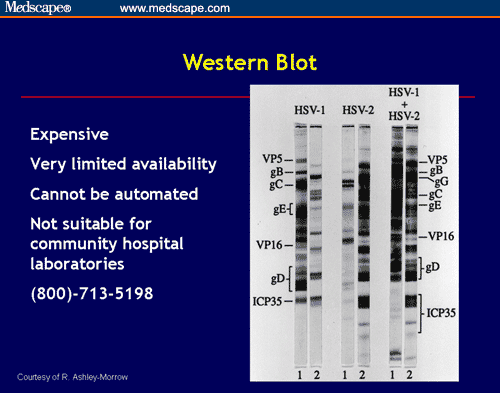 
The Focus kit showed a sensitivity of 93% and a specificity of 95%. The kit contains reagents for testing 24 samples. A colored precipitate forms if the anti-human conjugate has bound and the band(s) are interpreted. This assay involves two steps, 1) patient sera is diluted and incubated with the antigen test strips, 2) the blots were incubated with alkaline phosphatase-conjugated got anti-human IgG (Fc or heavy chain specific,) and then the BCIP/NBT substrate is added. The kit provides purified native and recombinant HSV antigens on a nitrocellulose membrane, which is dried and cut into strips. Clinical and Vaccine Immunology, 15(8):1301-1303., Compared to three commonly used commercial immunoassays (Focus, Kalon, BioKit) for detecting HSV-2 antibodies in China, Focus (Herpes Select) kits are available for HSV-1 and -2. Comparison of Three Commercial Immunoassays for Detection of Herpes Simplex Virus Type 2 Antibodies in Commercial Sex Workers in Yunnan Province, China. Sensitivity to a level near that of the Western blot.ĭISCUSSION: Ngo et al., 2008 1 Ngo, Thoai, D., Oliver Laeyendecker, Rhoda Ashley Morrow, Shenghan Lai, and Thomas C. Improvements in the EIA assay have increased its However, the WBA is expensive and technically challenging which limits its available in clinical settings. While the WBA was more sensitive in detecting HSV antibodies early in the primary course (within 40 days of infection) of HSV-1 infections, the EIA assay had similar sensitivities to the WBA assay for HSV-2 antibody detection within 40 days of infection (2). The sensitivity of this assay is primarily due to the detection of antibodies to more than one viral protein in the single assay. Of these, the Western blot assay (WBA) is still considered the “gold standard” in the detection of antibodies to viral proteins. The three most commonly used assays are Western blot assay (WBA), enzyme-linked immunosorbent assay (ELISA), and enzyme immnodot assays (EIA). INTRODUCTION: Various assays have been developed over the last thirty years for detecting Herpes virus antibodies in patient sera. Molecular virology techniques (particularly those using PCR) are likely to become the diagnostic methods of choice for both HSV infection and VZV infection once these tests become commercially available.By David Kilpatrick, PhD and Abbas Vafai, PhD 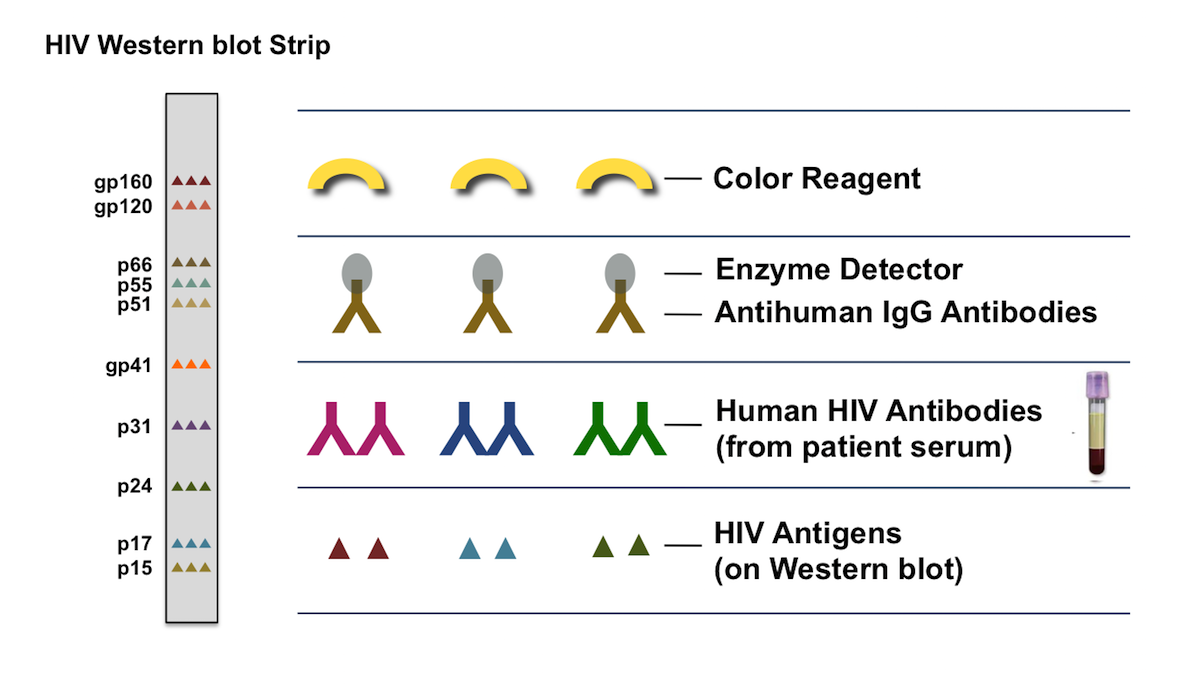
Immunomorphologic techniques have been useful adjuvant methods for both the diagnosis and the differentiation of HSV and VZV infections. The fluorescent antibody to membrane antigen test and viral tissue culture have been the principal methods for diagnosing VZV infection. The gold standard for establishing the diagnosis of HSV infection has been the viral tissue culture. Serologic tests traditionally do not have a major role in the diagnosis of HSV infection yet, new type-specific methods using Western blot assays may be useful for confirming the presence of unrecognized, subclinical HSV2 infections that are presently being underdiagnosed by current procedures. An expedient, slightly more expensive, reliable technique for establishing a HSV infection, yet not able to differentiate the subtype of that infection, is a recently marketed monoclonal antibody-based filtration type enzyme immunoassay (Kodak SureCell Herpes Test Kit). In the office or at the bedside of a hospitalized patient, a positive Tzanck smear preparation is an inexpensive, rapid, and morphologic technique for confirming a suspected diagnosis of a herpesvirus infection. Several laboratory diagnostic methods are available for the diagnosis, differentiation, and subtyping of HSV and VZV infections.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
